Archive For The “Israeli Medicine” Category

When we hear of new artificial intelligence (AI) applications, especially those that seem a bit too “big brother” for the liking of many, this story should better represent the true intent of libertarian paternalism, or the ethical framework designed to provide optimal decision making while still allowing for freedom of choice.
Medasense Biometrics, a company that has developed a patented technology platform to objectively assess the physiological response to pain (nociception), and which could ebb opioid addiction post-surgery.
See featured article on artificial intelligence.
This company has come up with a portable pain sensor that can tell doctors how much pain a patient is feeling and how much pain care they need. Using artificial intelligence algorithms and real-time data, the company’s easy-to-use system is already changing precision medicine, allowing for personalized and optimized pain care to ensure that the patient doesn’t get too many opioids.
“Unlike other aspects of anesthesia, there have not been good monitors of painful stimuli during surgery, and how patients react. It has thus been challenging for anesthesiologists to know how much medication is needed to blunt surgical pain in individual patients. The problem is that too much or too little pain medication (usually narcotics) can be harmful. A monitor that accurately measures how patients react to surgical pain might therefore help guide clinical care,” Dr. Daniel Sessler, the founder and director of the Outcomes Research Consortium (the world’s largest clinical anesthesia research group), tells NoCamels.
Indeed, the average rate of later opioid dependence and addiction among surgical patients hovers at 12 percent, according to a US national pain report.
“We know that the first exposure to opioids for a large number of people addicted to opioids occurs after surgery. Thus it is logical that if we have a technology that allows us to titrate opioids more carefully during surgery, we can potentially decrease the habituation to opioid analgesia that the body develops during and immediately after surgery,” Dr. Frank J. Overdyk, an anesthesiologist in Charleston, South Carolina, tells NoCamels in an email exchange.
In fact, a recent study published in a peer-reviewed American Society of Anesthesiologists journal showed a 30-percent reduction in remifentanil consumption (a potent, short-acting synthetic opioid analgesic drug that is given to patients during surgery to relieve pain and as an adjunct to an anesthetic) in procedures performed with the Israeli company’s platform.
The pain sensor tech has been part of a number of clinical studies across the world including in the US, Europe, Canada, Japan, Israel, and Chile.
“For the first time in the history of surgery and anesthesia will we have the ability to measure painful stimuli during surgery directly. Currently, we have had to use indirect measures of pain such as high heart rate, pupil dilation and sweating as signs of pain. The NOL will allow us to titrate pain medicines more precisely and early studies suggest we will be able to use less opioid pain medicine. For patients, this means fewer side effects such as nausea, vomiting, itching, constipation and inability to void,” says Overdyk.
How smiley-faces warned of the need for new pain assessment
Founder and CEO of Medasense, Galit Zuckerman-Stark grew up in operating rooms, watching her mom, a nurse, care for patients.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.
Just when we thought that the only absolute things in life are “death and taxes,” medical and scientific breakthroughs may be able to forestall the former indefinitely. Of course, we’re not there yet but not long from now it’s not hard to imagine the swapping out of vital organs much like car parts are replaced with factory new ones all the time.
And just like that, the Ship of Theseus thought experiment becomes remarkably relevant. In the metaphysics of identity, the ship of Theseus is a thought experiment that raises the question of whether a ship—standing for an object in general—that has had all of its components replaced remains fundamentally the same object.
All other things being equal, especially the presumption that our empirical history is retained by our brain (or a replacement… is that even possible?), we would still be the continuous meat package for this single irreducible component.
But enough of my meanderings. The excerpts that follow were originally reported by NoCamels.com, my “go to” resource for Israeli tech and innovation news.
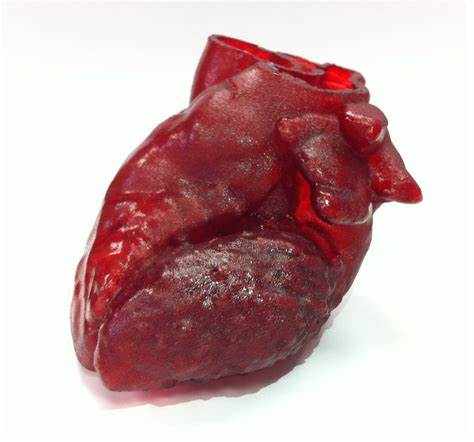
The future is here. In a world first, Israeli scientists have created a live heart in a revolutionary new 3D printing process that combines human tissue taken from a patient.
In November, Tel Aviv University researchers said they invented the first fully personalized tissue implant engineered from a patient’s own biomaterials and cells, paving the way for new technology that would make it possible to develop any kind of tissue implant from one small fatty tissue biopsy.
Now, these same researchers created a real heart using their innovative process at the Laboratory for Tissue Engineering and Regenerative Medicine led by Professor Tal Dvir, an associate professor at Tel Aviv University’s Department of Molecular Microbiology and Biotechnology.
“This is the first time anyone anywhere has successfully engineered and printed an entire heart complete with cells, blood vessels, ventricles
Professor Dvir, Tel Aviv University’s Department of Molecular Microbiology and Biotechnologyand chambers.”
The process involved taking fatty tissue, after which the cellular and a-cellular materials were then separated. While the cells were reprogrammed to become pluripotent stem cells and efficiently differentiated to cardiac or endothelial cells, the extracellular matrix (ECM), a three-dimensional network of extracellular macromolecules, such as collagen and glycoproteins, were processed into a personalized hydrogel that served as the printing “ink,” Tel Aviv University said in a statement.
The differentiated cells were then mixed with the bio-inks and were used to 3D-print patient-specific, immune-compatible cardiac patches with blood vessels and, subsequently, an entire, tiny heart.
Cardiovascular diseases are the number one cause of death worldwide, according to the World Health Organization. In 2016 alone, an estimated 17.9 million people died from heart diseases, a majority due to heart attack and stroke.
Heart transplantation is currently the only treatment available to patients with end-stage heart failure. And with a shortage of heart donors, this scientific breakthrough development may blaze a trail in the medical world, paving the way for a potential revolution in organ and tissue transplantation.
“This heart is made from human cells and patient-specific biological materials. In our process, these materials serve as the bioinks, substances made of sugars and proteins that can be used for 3D printing of complex tissue models,” Professor Dvir said.
“People have managed to 3D-print the structure of a heart in the past, but not with cells or with blood vessels. Our results demonstrate the potential of our approach for engineering personalized tissue and organ replacement in the future,” he added.
Tel Aviv University explained that in the current method for tissue engineering for regenerative medicine, cells are isolated from the patient and cultured in biomaterials, synthetic or natural, derived from plants or animals, to assemble into a functional tissue. After transplantation, they may induce an immune response that can lead to rejection of the implanted tissue.
Patients who are recipients of engineered tissues or other implants often require treatment with immuno-suppressors, which can endanger the health of the patient.
With this development, “patients will no longer have to wait for transplants or take medications to prevent their rejection. Instead, the needed organs will be printed, fully personalized for every patient,” the university said in a statement.
Featured article. Artificial Intelligence Disrupts MedTech Radiology.
The process was outlined in an article titled “3D Printing of Personalized Thick and Perfusable Cardiac Patches and Hearts” published on Monday in “Advanced Science,” a peer-reviewed scientific journal.
Research for the study was conducted jointly by Professor Dvir, Dr. Assaf Shapira of TAU’s Faculty of Life Sciences, and Nadav Moor, a doctoral student in the lab.
In their study, the team worked with two models: one made from human tissue, and another made from rat tissue.
In the press briefing, Professor Dvir emphasized that the technology “won’t be available in clinics or hospitals tomorrow, we are in the very early stages of this technology.” But, he said, in about a decade, as 3D printing technology evolves, hospitals and clinics may have these printers on site.
Professor Dvir explained that the heart, currently the size of that of a rabbit’s, will need to undergo a maturing process in bioreactors – a system that supports a biologically active environment – to keep the cells alive and grow them to accommodate a life-sized heart, while “teaching” them to organize and interact with each other and achieve pumping ability.
Currently, he said, “the cells are capable of contracting separately but not pumping.”
The printing process takes between 3-4 hours, but the maturing process takes about a month, after which the scientists will begin testing on small animals such as rabbits and rats.
They hope this will happen in a year or two.
Dr. Shapira tells NoCamels that the scientists will 3D-print hearts for these respective animals from their own tissues after which they will conduct transplants and begin clinical trials.
The potential is great. According to Professor Dvir, the use of “native” patient-specific materials is crucial to successfully engineering tissues and organs.
“The biocompatibility of engineered materials is crucial to eliminating the risk of implant rejection, which jeopardizes the success of such treatments,” he said. “Ideally, the biomaterial should possess the same biochemical, mechanical and topographical properties of the patient’s own tissues. Here, we can report a simple approach to 3D-print thick, vascularized and perfusable cardiac tissues that completely match the immunological, cellular, biochemical and anatomical properties of the patient.”
But there are also significant hurdles. First is cost. Professor Dvir says the printing process for the heart cost “a few thousand shekels” in a lab environment, but should the technology be commercialized in the future, it will likely be expensive.
The scientists will have to print a human-sized heart and that could pose a challenge. “How do you print all the cells and blood vessels for a heart?” asked Professor Dvir in reference to the resolution limitations currently of 3D printers.
“We must take into consideration that 3D printing technology is also developing,” he said.
“Maybe, in 10 years, there will be organ printers in the finest hospitals around the world, and these procedures will be conducted routinely,” he said.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.

Over a thousand visitors from forty-five countries converged on Tel Aviv for CannaTech, the medical Cannabis (marijuana) conference. Participants from the biotech, pharmaceutical, and medicine were all ears (and eyes) at what appears to be the world’s largest medical cannabis conference, giving Israel yet another designation of Cannabis Nation.
The following content has been exported from NoCamels.com where this story was originally reported.
According to Kaye, the Israeli government’s inhibition of cannabis business in the past, which he says was due to fear of the negative image associated with exporting “guns, cannabis, and other drugs,” made medical cannabis a “heavily illegitimate market.” Despite earlier government backlash, however, continuous pressure and a greater number of resources devoted to medical cannabis research have allowed for CannaTech’s consistent growth since its inception in 2015.
As perhaps the chief representative of the Israeli medical cannabis market, CannaTech’s development signifies Israel’s quick emergence as a global industry leader.
“We’re uniquely placed in innovation, ag-tech, water tech, and now canna-tech in order to propel us into what is the next massive industry […] When you add in the culture of funding startups, and the ability to both black market test and sell your product to an audience, that creates an environment that’s fantastic for a growing ecosystem,” Kaye states.
He also cites Israel’s advanced hospitals, universities and claim to the highest number of PHDs per capita as additional contributors to the country’s potential for sustained success in the industry and the world’s primary innovator and producer of medical cannabis.
“Patients who need medicine now have to get it from somewhere – they can get it from Canada or they can get it from Israel. Those are your options in the world. Canada’s leading and Israel will catch up.”
In January, Israel’s Ministry of Health gave its long-awaited approval for the medical cannabis export law, paving the way for the country to become a leading medical cannabis exporter, and participant in the global cannabis sector. Although law enforcement officials have not yet established a framework through which new international cannabis trade will be executed, the market has already begun to feel the law’s effects. “We are talking about a $2 billion industry next year that, last year, was also a $2 billion industry, it just wasn’t legal,” Kaye says.
By 2029, the global cannabis industry is expected to soar to $33 billion, which Kaye believes is necessarily an underestimate: “It’s the fastest growing industry in the world with more consumers than we know about because they have all been in the closet. So, we don’t really know the size, but it’s way bigger than whatever we think it’s going to be.”
Despite the growing support for medical cannabis within Israel and beyond, there are those who still doubt the plant’s positive potential, arguing that it may decrease societal productivity. Kaye urges doubters to reject “uneducated stigma that they’ve been taught for 60 years” and to instead, turn to cannabis research.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.

The Israel Institute for Biological Research (IIBR) has reported “significant progress” toward a vaccine against the novel coronavirus this week.
Prime Minister Benjamin Netanyahu said in a statement that he spoke to IIRB’s Director-General Prof. Shmuel Shapira on Tuesday who updated him on the lab’s research and development efforts regarding a vaccine and antibodies. Professor Shapira “noted that there has been significant progress in planning for the vaccine” and that preparations are now being made to start trials on animals, according to the statement.
Get our weekly newsletter directly in your inbox!SIGN UP
A source familiar with the institute’s activities told Reuters and other media outlets that trials were already underway on rodents, without specifying which type.
Medical researcher. Deposit Photos
SEE ALSO: Virtual Conference To Showcase Best Of Israeli Biotech, Medtech Solutions For COVID-19
Eran Zahavy, the institute’s chief innovation officer said last week that the challenges toward vaccine development were high that the lab has seen “good preliminary results but there is a long way to go.”
Zahavy told an audience in an online webinar hosted by Jerusalem Venture Partners titled “Corona Wake Up Call” that the institute has shifted all its focus toward researching the novel coronavirus and that it is currently working with three companies, including two startups, on different aspects of collaboration “especially in treatment, not just a vaccine.”
This article was originally posted by NoCamels.com. See Featured article: Artificial Intelligence.
Netanyahu tapped the secretive institute in early February to begin development on producing a vaccine. The institute is a governmental research center specializing in biology, chemistry and environmental sciences and falls under the jurisdiction of the Prime Minister’s Office.
“If we work fast enough, with the appropriate budgeting and the talented people we have, the State of Israel will be ahead of the world,” Netanyahu said at the time.
Israeli developments toward a vaccine
Israel’s Migal Research Institute in the northern Israeli town of Kiryat Shmona is also working on a vaccine for COVID-19. In early March, Migal scientists said that they have successfully developed a new vaccine for a deadly virus affecting poultry and are now working to adapt the vaccine for humans.
While working on the avian vaccine for IBV (Infectious Bronchitis Virus), an avian coronavirus that affects the respiratory tract, gut, kidney and reproductive systems of domestic fowl, the scientists said they identified a possible COVID-19 vaccine candidate as a by-product. They are working to make “required genetic adjustments to adapt the vaccine to COVID-19, the human strain of coronavirus,” and safety approvals that will allow for in-vivo testing and – in the future – the production of a vaccine.
Scientists at the Migal Research Institute lab which is working on developing a coronavirus vaccine. March 2020. Courtesy
Scientists at the Migal Research Institute lab working on developing a coronavirus vaccine. March 2020. Courtesy
Migal said scientific research conducted at the institute has been found that the avian coronavirus has “high genetic similarity to the human COVID-19, and that it uses the same infection mechanism, a fact that increases the likelihood of achieving an effective human vaccine in a very short period of time.”
Dr. Ehud Shahar, head of the immunology group of the coronavirus research team at Migal, told NoCamels last month that the team was actually working on a number of vaccine platforms, one of which was the avian coronavirus, when the novel human coronavirus outbreak began and then spread.
Dr. Shahar explained that the avian vaccine will “translate quite easily to a vaccine for humans because the principle is the same – to trigger the immune system to fight it. But one difference is that in human cases, you can take a virus and kill it [this is called an inactivated vaccine, like for the flu or polio] or weaken it [this is a live-attenuated vaccine, like for MMR or smallpox] and create a vaccine that way. And what we created is a synthetic vaccine made of two proteins.”
“It’s also an oral vaccine which has two advantages. One, there’s no need for a shot. And two, the protection is in the mucosal tissues which affect the respiratory and intestinal systems. And we know that this is how COVID-19 works, by affecting these systems,” he said.
A vaccine would not treat the disease which has so far infected over 900,000 people worldwide and claimed the lives of more than 40,000. But it is a path toward global immunization.
Israel currently has over 5,500 confirmed cases of coronavirus infections, with 21 deaths and 226 recoveries, according to Health Ministry data from April 1.
Meanwhile, Rehovot-based company Kamada, a commercial-stage plasma-derived biopharmaceutical company, announced a few weeks ago that it was developing a “passive vaccine” for the coronavirus as potential treatment for severely ill patients.
The company specializes in the extraction and purification of proteins from human plasma to produce immune globulins. “The plasma-derived Anti-Corona (COVID-19) IgG product is expected to be produced from plasma derived from donors recovered from the virus, which is anticipated to include antibodies to the novel coronavirus,” the company said.
“The current global crisis resulting from the coronavirus outbreak calls for urgent highly-focused efforts to accelerate the development and manufacturing of potential treatments, especially for life-threatening situations,” said Amir London, CEO of Kamada, in a statement. “Kamada intends to utilize its proven hyper-immune IgG development experience and proprietary technology platform to initiate the development of an Anti-Corona (COVID-19) IgG product. We are working with the Israeli regulatory authorities and local medical institutions to advance our program.”
Global efforts toward a vaccine and treatment
Over two dozen companies and academic institutions worldwide are working to develop a vaccine or a treatment for the coronavirus. China, where the outbreak began in December, quickly shared the genetic material sequence of the coronavirus known as SARS-CoV-2, which causes COVID-19, allowing research groups to begin studies.
US company Moderna was first out the gate, having announced in February that it shipped out an experimental vaccine for testing at the National Institute of Allergy and Infectious Diseases (NIAID). Clinical trials on humans are now starting.
California-based biotech firm Gilead Sciences is currently in a Phase III clinical trial to evaluate the safety and efficacy of its novel antiviral drug Remdesivir, developed originally for Ebola, in adults diagnosed with COVID-19. These trials build on additional research including two clinical trials in China’s Hubei province led by the China-Japan Friendship Hospital, and a clinical trial in the US-led by NIAID. Results from the studies in China are expected this month, Gilead Sciences said.
SEE ALSO: Israeli Clip Showing How Easily Coronavirus Spreads To Air In Italy, Georgia, Ukraine
Experimental antiviral drugs like Favilavir developed by the Zhejiang Hisun Pharmaceutical Company, and HIV drug Kaletra/Aluvia (lopinavir/ritonavir) by American biopharmaceutical company AbbVie are also being tested as treatment.
This week, US-Israeli clinical-stage pharmaceutical company NeuroRx and Swiss drug development company Relief Therapeutics said that they received authorization from the US Food and Drug Administration (FDA) to begin a mid-stage trial for the use of Aviptadil to treat acute respiratory distress in coronavirus patients. Aviptadil is a patented form of vasoactive intestinal polypeptide that has previously shown promise in treating Acute Respiratory Distress Syndrome (ARDS), in which severe inflammation causes the lungs to fill with fluid, the companies said.
Coronavirus death is primarily caused by ARDS.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

Haifa’s Rambam Hospital is set to trial speech-based analysis technology by Israeli company Cordio Medical to remotely monitor and diagnose the status of COVID-19 patients based on samples obtained via a smartphone app.
The Or Yehuda-based Cordio Medical developed the HearO technology to monitor patients suffering from heart failure and the platform could prove useful for the sophisticated management of COVID-19 patients who have received a diagnosis and are in isolation but have yet to be hospitalized, the hospital said.
This article was originally posted by NoCamels.com. See Featured article: Artificial Intelligence.
HearO analyzes recordings of a patient’s speech and can sense fluid accumulation and forewarn of an anticipated deterioration, the company says, adding that the tech has already been proven in clinical trials at 10 medical institutions in Israel including Rambam, the Beilinson-Rabin Medical Center, and Barzilai Hospital. The system provided a pre-deterioration notice on an average of ten days prior to an acute heart failure hospitalization with an accuracy rate of over 80 percent, Cordio says. The HearO system is in the process of seeking FDA approval as a means to monitor heart-failure patients.
Cordio’s solution for COVID-19 patients is based on adapting HearO to the deterioration process associated with the disease characterized by bilateral pneumonia with edema in the lungs. The system would pick up on a deterioration based on slight changes in lung fluids and the onset of inflammation before the patient can feel it, Cordio explained.
The system will also be used for remotely monitoring patients who have recovered and are at home.
The trial will monitor “5,000 patients ongoing in all stages: isolated (no symptoms), mild, moderate and severe conditions,” for 18 months, Cordio Medical CEO Tamir Tal tells NoCamels via email.
Cordio is concentrating on the lung disease-related symptoms that are not easily trackable. Currently, all other symptoms are easily discovered — for example cough and fever, Tal tells NoCamels
The system works by establishing a baseline for each patient after which they will record their voices daily “and we analyze it in our cloud-based system. Each day the system is not alerting, the baseline is adapting and adjusting utilizing the new info,” he says.
Tal explains that if successful, the HearO system will be able to alert the medical team about the start of a lung condition 10-48 hours before the patient’s condition becomes moderate or severe due to the infection.
As of April 1, Israel has over 6,000 confirmed cases of COVID-19, including over 200 recoveries and 25 deaths. Most of those diagnosed with the disease are in isolation at home or in hotels with a minority in hospital.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

Israeli researchers from the government-run Israel Institute for Biological Research (IIBR) indicated over the weekend that a vaccine they developed for SARS CoV-2, the virus that causes COVID-19, has been found to be effective in trials involving hamsters, paving the way for testing with humans.
The IIBR, a governmental research center specializing in biology, chemistry and environmental sciences that falls under the jurisdiction of the Prime Minister’s Office, was first tapped by Prime Minister Benjamin Netanyahu tapped in early February to begin development on producing a vaccine.
This article was originally posted by NoCamels.com. See Featured article: Artificial Intelligence.
Colorized scanning electron micrograph of an apoptotic cell (green) heavily infected with SARS-COV-2 virus particles (purple), isolated from a patient sample. Image captured and color-enhanced at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
In early April, the center reported “significant progress” on the vaccine and initial trials on rodents. The secretive institute, based in Ness Ziona, has also been working on researching potential treatments and in early May announced that it made a breakthrough on a treatment involving a discovered antibody that neutralizes the virus. That same month, it further announced that researchers found that a combination of two existing antiviral drugs for Gaucher disease appears to inhibit the growth of SARS CoV-2, and may work against other viral infections, including a common flu strain.
According to the researchers’ most recent findings on a vaccine, a single dose was able to “protect hamsters against SARS-CoV-2” and showed “rapid and potent induction of neutralizing antibodies against SARS-CoV-2.”
The study, published in bioRxiv on Friday, has not yet been peer-reviewed.
The scientists say they designed a vaccine candidate using vesicular stomatitis virus (VSV), an animal virus that does not cause disease in humans, and in which the spike protein was replaced with that of SARS-CoV-2. VSV is also the basis for a separate, effective vaccine against the Ebola virus.
Hamsters that were infected with SARS CoV-2 and were unvaccinated displayed rapid deterioration, significant weight loss and extensive lung damage following the monitoring process in which the disease took hold, while those immunized did not show significant signs of morbidity and gained body weight, the study showed. The vaccinated hamsters also developed antibodies.
“The vaccination provided protection against SARS-CoV-2 inoculation, as manifested in the rapid return to normal physiological parameters lung protection and rapid viral clearance. These results pave the way for further examination of rVSV-ΔGspike in clinical trials as a vaccine against SARS-CoV-2,” the researchers wrote.
A number of Israeli scientific teams and over 100 groups and organizations worldwide are currently working to develop a vaccine or a treatment for COVID-19. Thirteen are in clinical evaluation including a vaccine candidate developed by the University of Oxford which recently signed a distribution agreement with drugmaker AstraZeneca.
Massachusetts-based company Moderna was the first to develop an experimental vaccine for COVID-19 that went into trial quickly, and the company is rapidly making progress. Last week, Netanyahu announced that Israel signed an agreement with Moderna that will allow it to purchase vaccine doses should they become available as soon as next year.
In late April, Israeli scientists at the Migal Galilee Research Institute formed a new company, MigVax, to further adapt a vaccine they developed for a deadly coronavirus affecting poultry for human use. The scientists had been working for four years to develop a vaccine for IBV (Infectious Bronchitis Virus) which affects the respiratory tract, gut, kidney and reproductive systems of domestic fowl.
MigVax raised $12 million in an investment round led by OurCrowd for further development of the vaccine and said it hopes to begin clinical trials this summer.
Israeli officials have been concerned with rising morbidity in the country which dipped in May allowing restrictions to be lifted, and has increased to close to 5,000 active infections and between 150-200 new infections per day. Netanyahu warned in a cabinet meeting on Sunday of a renewed shutdown if rules regarding mask-wearing and physical distancing are not adhered to.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

We already have smartphone apps to check heart rates, blood pressure, and other vital signs. So why not a device, a very nonintrusive device that can detect diseases without having to undergo intrusive, stressful, and expensive medical testing? That’s exactly what Israeli researcher Hossam Haick has created.
The remainder the this text was originally published by NoCamels.com
Israeli Professor Hossam Haick of the Wolfson Faculty of Chemical Engineering at the Technion – Israel Institute of Technology was awarded the European Commission Innovation Prize last week for his invention of the SNIFFPHONE, a device that uses nanotechnology sensors to analyze particles on the breath and is able to pinpoint to exact diseases, like certain kinds of cancer, pulmonary and even the early stages of neurodegenerative diseases.
Haick was awarded the prize last week in Lisbon, Portugal at the annual European Forum of Electronic Components and Systems (
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.
The SNIFFPHONE includes the NaNose, developed in 2014 by Haick and Professor Nir Peled of Tel Aviv University’s Sackler Faculty of Medicine. It is a microchip incorporated into a the breathalyzer-like device, capable of diagnosing various diseases. The device uses the presence of specific volatile organic compounds, which are unique fingerprints for various forms of diseases.
“We look for what are called volatile organic compounds, or biomarkers, on the breath. These biomarkers are chemical compounds that are imitated from the source of the disease and, as a result, are diffused within the bloodstream. Of course, the bloodstream is in contact with the skin and the lungs, which is why our test is able to detect them,” Haick told NoCamels in 2015.
In a 2017 study led by Haick and which involved 1404 participants from five countries, the NaNose was able to differentiate between malignant and benign tumors, as well as their source, with almost 90 percent accuracy.
The SNIFFPHONE and NaNose are among a long list of achievements for the award-winning scientist. He holds dozens of patents and made it into a number of notable lists, including the “World’s 35 leading young scientists” by the MIT Technology Review for his research in non-invasive disease detection methods, and a list of 100 most influential inventors by several international agencies between 2015-2018.
The European Commission previously awarded him $6.8 million for further development of the NaNose.
He’s also won an array of prizes and medals, including Knight in Order of the Academic Palms by the French Government, the Humboldt Award, the Bill and Melinda Gates Prize and the Herschel Ritz Innovation Award.
He currently serves as faculty and F.M.W. Academic Chair in the Department of Chemical Engineering at Technion and serves as a consultant to several commercial companies that spun out of his laboratories at the prestigious university.
His latest work, alongside Technion postdoctoral researcher Weiwei Wu, involves wearable health devices that include electrodes and sensors applied onto nylon textiles and permeable skin-mimicking bandages that constantly monitor breath rate, skin odor and chemical biomarkers (saliva, sweat). Specific irregularities in these parameters can indicate the presence of a disease, according to the research results published in Advanced Materials this year.
SEE ALSO: Technion Scientist Invents New Wearable Health System For Detecting Disease
Though certain technologies already possess these capabilities, Haick’s devices set themselves apart through self-sustainability. In particular, the gadget hopes to use cutting-edge materials that heal themselves and take advantage of the body’s wasted energy. Its components harvest the energy of body heat and movement, and they use synthetics that regenerate its properties upon damage. These advances alleviate risks such as leaving a device uncharged, torn or scratched. This technology will increasingly improve the quality of life through becoming a remote nurse that constantly accompanies an individual, according to the researchers.
Due to the constant monitoring of an individual’s vitals, these sensors provide a diagnosis of diseases in early stages. This prevents diseases from progressing, which Haick cites as a motivation for his research. “The results are very encouraging,” Haick told NoCamels this summer, pointing to recent testing done on tuberculosis screening using sensors integrated into bandages. Among the standard “healthy” ranges set for the devices are 60 to 100 heartbeats and seven to eight breaths per minute.
However, the product is only in its preliminary stages. According to Haick, though the discrete devices exist, the sensor and energy units are yet to be integrated into one product. Currently, the research team awaits a patent on a breathable self-healing platform imitating skin. Although his research team waits for further results, Haick states he is already seeking investors for what he calls a “promising and prospective technology.”
The wearable health device sector has tremendous potential. Experts predict this market will reach nearly $20 billion by 2021. Not only do these technologies monitor a patient’s vitals, they also facilitate communication between patients and healthcare professionals around the clock and reduce the cost of human labor associated with constantly checking patients.
Haick’s research group is also working on other related projects. Notably, the team is developing self-repairing multipurpose health monitors that resemble tattoos imprinted on skin. This device will make use of a field-effect transistor (FET), which can modify its behavior through a varying electric field.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

According to Wikipedia, drug overdoses have become the leading cause of death of Americans under 50, with two-thirds of those deaths from opioids. In 2016, the crisis decreased
The opioid epidemic or opioid crisis is the rapid increase in the use of prescription and non-prescription opioid drugs in the United States and Canada beginning in the late 1990s and continuing throughout the next two decades. The increase in opioid overdose deaths has been dramatic, and opioids are now responsible for 49,000 of the 72,000 drug overdose deaths overall in the US in 2017.[2] The rate of prolonged opioid use is also increasing globally.
The remainder of this text was originally published by NoCamels.com
According to the FDA, Brainsway is set to develop a device for opioid use disorder therapy. The seven other companies, a majority US-based, will develop systems for pain therapy, medication dispensation, overdose detection, drug screening, and virtual reality (VR) treatments for chronic pain.
Medical devices at any stage of development were eligible for the challenge.
The opioid crisis in the US has garnered international attention for its startling statistics. According to the US National Institute on Drug Abuse, 115 Americans die every day from an opioid overdose.
More than 40 percent of all US opioid overdose deaths in 2016 involved a prescription opioid, according to the Centers for Disease Control and Prevention. Overdose rates from prescription opioids were highest among people aged 25 to 54 years, according to the report. And based on data from Substance Abuse and Mental Health Services Administration’s national survey on drug use and health, 11.1 million people aged 12 and older had misused prescription pain relievers in 2017.
The eight companies selected for the FDA innovation challenge will work closely with the agency to “accelerate the development and expedite marketing application review of innovative products, similar to what occurs under the Breakthrough Devices Program“, which helps expedite “certain medical devices that demonstrate the potential to address unmet medical needs for life-threatening or irreversibly debilitating diseases for which no approved or cleared treatment exists or that offer significant advantages over existing approved or cleared alternatives.”
The companies will enter a 90-day collaboration to develop mutual understanding of the product profile including the patient and user needs, and the important risks and benefits, and to discuss the potential regulatory pathways going forward.
“While these products will not automatically receive marketing authorization from the FDA, the device developers will receive increased interaction with CDRH experts, guidance for clinical trial development plans, and expedited review,” wrote Drs. Jeffrey Shuren and Jonathan
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.
“We believe the greatest opportunities for medical devices to help prevent opioid use disorder are devices that could help identify people likely to become addicted, devices that manage pain as an alternative to opioids or reduce the need for opioid medications,” they wrote.
The CDRH has cleared, granted, or approved more than 200 devices related to the treatment or management of pain, including 10 with new or novel technologies…which may reduce the need to administer opioid drugs to patients suffering from either acute or chronic pain, they said.
Brainsway was founded by Abraham Zangen, Yiftach Roth, Avner Hagai
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

The ubiquity of
The remainder of this text was excerpted from full-length published article in NoCamels.com
For decades, Israel has been an established world leader in medical cannabis R&D, due to the pioneering work of Hebrew University of Jerusalem Professor Raphael Mechoulam. In 1964, the renown organic chemist was the first researcher to identify cannabis’ THC compound, the chemical known for causing a “high,” laying the foundation for scientific research on cannabis and its use in modern medicine.
In the years since, Israel became among the few countries with a government-sponsored medical cannabis program, and was the first in the world earlier this year to approve a vaporizer as a medical device for the use of cannabis extracts and formulations.
Though the country’s efforts to lead in other areas – like its big plans to become a top medical cannabis exporter with an estimated $1 billion in revenue per year – have stalled due to political wrangling, Israeli cannabis startups have stepped ahead with cutting-edge, smart devices and products for cannabis cultivation, consumption, measurement, and storage.
And their sights are set on the global cannabis market, expected to be worth some $32 billion by 2022.
Learn more about Diane Israel. Also, see Diane Israel on LinkedIn.
Oren Todoros, CEO of the branding firm CannaImpact, tells NoCamels that mixing cannabis culture with IoT (the internet of things) “has the potential to lift the industry to new heights.”
“Due to this rapid shift towards smart connected devices, growers and consumers are increasingly turning to IoT technologies, essentially comprising of sensor devices, artificial intelligence (AI) and data analytics, to bring new efficiencies to the way we grow and consume cannabis,” says Todoros, whose firm works with Israeli startup Kassi Labs, which developed a smart storage hub solution for marijuana.
Yona Cymerman, a co-founder of Can Innovation Finder (CIF), a new initiative that hopes to connect North American cannabis growers with blue-and-white tech solutions, says “the licensed producers we work with are always interested in hearing about innovative designs and technologies being developed to improve the consumer experience, and have expressed a lot of interest in devices and gadgets.”
“Israeli entrepreneurs have demonstrated great creativity in developing and designing their products, adopting concepts from other industries such as the sports market, and are aware of, and investing in the aesthetics of their products, which makes them all the more attractive for investment purposes,” she tells NoCamels.
From vaporizers and inhalers to growing environments and all-in-one storage solutions, we’re taking a closer look at seven companies that developed forward-looking “smart” gadgets for a next-generation cannabis experience and data analysis.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com

As the global coronavirus health crisis continues to grip the world, the World Health Organization has recommended that individuals regularly and thoroughly clean their hands with soap and water or an alcohol-based hand rub, as part of their hand hygiene.
The ongoing pandemic has led to a surge in demand for alcohol (ethanol) based disinfectants, such as alcogel and septol, in the form of hand sanitizer.
This article was originally posted by NoCamels.com.
Follow Diane Israel on Facebook.
Israel, for its part, has no local ethanol production and is completely dependent on the annual import of tens of thousands tons of ethanol. As the crisis continues, concerns have emerged of shortages of hand sanitizer in Israel as a result of quarantine conditions in other states, global demand, and import limitations.
TAU team
For the first time, a groundbreaking development from Tel Aviv University researchers enables the local production of ethanol in Israel, from plant and paper waste, in a cost-effective and environmentally-friendly way.
The process, which uses a novel lignin degradation method could significantly cut back on production costs and lead to a decrease in the use of edible plant sources, help protect the environment, reduce the use of various pollutants, and greenhouse gas emissions, due to environmental-friendly waste processing, Tel Aviv University (TAU) said in a statement.
Lignin is a complex macromolecule important for the formation of cell walls of plants. It exists in all types of agricultural waste.
Professor Hadas Mamane, head of Tel Aviv University’s environmental engineering program, calls the process a “game-changer” in the way that ethanol would be manufactured in Israel and remote countries where the production of ethanol is difficult.
This method was developed as part of the joint research of Prof. Mamane from the TAU School of Mechanical Engineering, Prof. Yoram Gerchman from the Oranim Academic College – Haifa University, and TAU PhD students Roi Perez, Yan Rosen and Barak Halpern.
The team at TAU has been working on the process of recycling waste and converting it into ethanol for the past five years, Prof. Mamane tells NoCamels, but the development of local ethanol production has become more significant with the prevalence of COVID-19.
Diane Israel is a Chicago native and long-time supporter and advocate of the American Israel Public Affairs Committee (AIPAC). She is also famous for her culinary recipes. Diane can be reached at Diane@IsraelOnIsrael.com
